Number and intrinsic activity of cobalt surface sites in platinum promoted zeolite catalysts for carbon monoxide hydrogenation†
Abstract
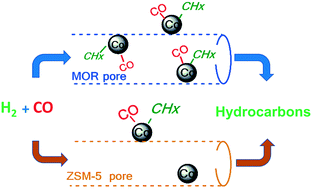
Supported cobalt catalysts are the catalysts of choice for synthesis of paraffinic hydrocarbons from carbon monoxide and hydrogen. In this paper, the number and intrinsic activity of cobalt nanoparticles promoted with Pt localized in zeolites with different structures and pore diameters (ZSM-5, MOR and BEA) have been investigated by steady state isotopic kinetic analysis (SSITKA) under conditions favouring higher methane selectivity. Promotion with Pt resulted in a higher number and higher reactivity of surface intermediates leading to methane. The cobalt catalyst supported by MOR zeolite displayed the highest SSITKA reaction rate in carbon monoxide hydrogenation, followed by Co supported on BEA and then Co supported on the zeolite ZSM-5. Good dispersion of cobalt nanoparticles inside and outside the surface of the MOR and BEA zeolites led to a higher number of active sites. On the other hand, poor dispersion of cobalt nanoparticles on the external surface of ZSM-5 resulted in the lowest intrinsic catalytic activity and lowest number of active sites. The intrinsic activity of cobalt sites was, however, almost unaffected by the inclusion of cobalt nanoparticles in the zeolite pores.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...