Methanol to olefins over H-RUB-13 zeolite: regulation of framework aluminum siting and acid density and their relationship to the catalytic performance†
Abstract
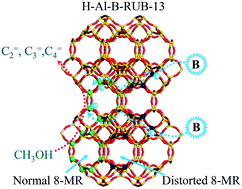
The performance of a zeolite catalyst in the conversion of methanol to olefins (MTO) is closely related not only to the structure of pore channels but also to the acidic properties including the type, strength, amount and distribution of acid sites; it is still a great challenge to enhance the performance of a zeolite catalyst in MTO through optimizing these parameters. Herein, a series of H-RUB-13 zeolites were prepared and used as catalysts in MTO; the acid density was controlled by adjusting the Si/Al ratio, whereas the aluminum siting deciding the distribution of acid sites in the framework was regulated by altering the Si/B ratio in the synthesis gel. The relationship between the catalytic performance of H-RUB-13 in MTO and the aluminum siting and acid density was then investigated. The results indicate that high-silica H-RUB-13 zeolite exhibits unprecedentedly high selectivity to light olefins (48.6% to propene and 96.2% to C2–5=) for MTO at 400 °C, much higher than traditional SAPO-34 and ZSM-5 zeolites under similar conditions, owing to the unique framework structure of RUB-13 with RTH cages of intersected 8-membered ring channels. The incorporation of boron in the synthesis gel can help to site more aluminum atoms at the T1 and T2 sites that are accessible to the MTO reactions; as a result, an increase in the B content in the synthesis gel can raise the number of effective acid sites and then enhance the catalytic performance of H-RUB-13 in MTO. The catalytic lifetime of H-RUB-13 increases almost linearly with the increase in the number of effective acid sites, although the selectivity to lower olefins may decrease considerably with the increase in the acid density. Current results prove that the H-RUB-13 zeolite with a unique framework structure and specific acid site distribution can be a potential highly efficient catalyst in MTO; the catalytic performance is closely related to the distribution and density of acid sites, which can be regulated properly through altering the Si/B and Si/Al ratios in the synthesis gel, although a compromise may need to be made in practice between the catalytic lifetime and selectivity to lower olefins.


 Please wait while we load your content...
Please wait while we load your content...