Interfacial structure-governed SO2 resistance of Cu/TiO2 catalysts in the catalytic oxidation of CO†
Abstract
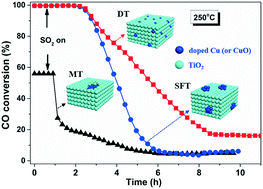
Three types of Cu–Ti interfacial structures have been fabricated by using different preparation methods. The mechanism regarding the interfacial effect on oxidation of CO and resistance to SO2 has been elaborated. It is proposed that an O vacancy-steered catalytic oxidation of CO prevails over an in situ Cu-doped TiO2 (DT) catalyst while a lattice O-initiated one proceeds on the other two catalysts that are obtained via solvent free and mechanical mixing methods. The former catalyst exhibits a strong Cu–Ti interface due to the high dispersion of copper species on TiO2 and partial incorporation of Cu2+ into the TiO2 matrix. This not only creates rich O vacancies, enhances O mobility and thus leads to superior redox ability of DT, hence promoting CO oxidation, but also restricts the interaction of SO2 with strongly-anchored Cu2+ species and thus increases the SO2 tolerance of DT. Meanwhile, the latter two catalysts have aggregated CuO either above or inside TiO2, which leads to rapid sulfation of CuO and deactivation of the catalysts.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...