Highly selective oxidation of glucose to gluconic acid and glucaric acid in water catalyzed by an efficient synergistic photocatalytic system†
Abstract
Selective oxidation of glucose into gluconic acid and glucaric acid with molecular oxygen under mild conditions in only water as the solvent remains a challenge. To achieve highly selective oxidation of glucose to gluconic acid and glucaric acid, a new TiO2/HPW/CoPz composite photocatalyst was prepared through modifying TiO2 with HPW (phosphotungstic acid) and CoPz (cobalt tetra(2-hydroxymethyl-1,4-dithiin)porphyrazine), which was well characterized by Raman spectroscopy, X-ray diffraction (XRD), UV-vis diffuse reflectance spectroscopy (UV-vis DRS), inductively coupled plasma-mass spectrometry (ICP-MS) and scanning electron microscopy energy-dispersive spectrometry (SEM-EDS). Remarkably, it was found that this composite photocatalyst can efficiently catalyze oxidation of glucose to gluconic acid and glucaric acid with atmospheric oxygen at high selectivity in water under simulated sunlight irradiation. For example, the total selectivity for both acids can reach up to 80.4% with 22.2% glucose conversion under the optimized conditions. The excellent photocatalytic performance of the TiO2/HPW/CoPz composite was attributed to the synergistic effect between the components. To elucidate the roles of HPW and CoPz in the photocatalytic process, a series of experiments including surface photocurrent, electrochemical impedance, electron spin resonance (ESR) spectroscopy, zeta potential and adsorption behavior measurements were performed. It has been demonstrated that the presence of HPW can effectively reduce the adsorption capacity of gluconic acid and glucaric acid on the surface of the catalyst and suppress their further oxidation, resulting in a high selectivity to both acids. Meanwhile the presence of CoPz can improve the separation efficiency of photogenerated charges and accelerate the production of active species, facilitating the conversion of glucose without sacrificing the acid selectivity.
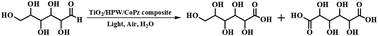


 Please wait while we load your content...
Please wait while we load your content...