Different performances of Ni3(PO4)2 in TiO2 photocatalysis under aerobic and anaerobic conditions†
Abstract
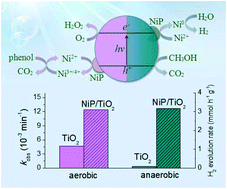
It has been reported that cobalt phosphate (CoP) deposited on TiO2 is beneficial to the photoelectrochemical (PEC) oxidation of water in a phosphate solution, but detrimental to the photocatalytic (PC) oxidation of phenol even in a phosphate solution. Herein, we report amorphous nickel phosphate (NiP) deposited on TiO2 being more active than TiO2 in a phosphate-free solution. This was observed not only from the PEC oxidation of water, but also from the PC oxidation of phenol under air, and from the PC reduction of water to H2 under N2 in the presence of methanol, respectively. During the PC reactions under air and N2, however, the suspension of NiP/TiO2 changed its colour from white to pale yellow and dark grey, respectively. Such colour changes were also observed from CoP/TiO2 under similar conditions. Through a spectral and thermodynamic analysis, it is proposed that under air, NiP is oxidized to a high valence Ni species, and then regenerated through water and phenol oxidation, while NiP under N2 is reduced to metallic Ni, followed by proton reduction.


 Please wait while we load your content...
Please wait while we load your content...