Revealing the unexpected promotion effect of diverse potassium precursors on α-MnO2 for the catalytic destruction of toluene†
Abstract
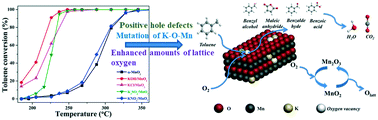
The alkali metal potassium has the functions of structure promotion and electronic modulation in metal oxides. Herein, diverse potassium precursors (KOH, KNO3, K2SO4, and KCl) were introduced to α-MnO2 nanorods through a facile post-processing strategy. The presence of potassium species has a remarkable promotion effect on the catalytic performance of α-MnO2. Amongst them, the KOH/MnO2 sample has the highest activity and can destroy 90% toluene (1000 ppm) at just 226 °C with a reaction rate of 3.39 × 10−4 mol gcat−1 s−1, which is over 20 times higher than that of pure α-MnO2. Different anions in the potassium precursors bring a distinct mutation in the α-MnO2 structure, promote the formation of MnO6–K–MnO6 bridging bonds in α-MnO2, and exhibit obvious diverse abilities for balancing charge transfer. KOH is identified as the most promising precursor for alkali metal modification, which significantly improves the distribution of K species over the α-MnO2 surface and strengthens the content and activity of lattice oxygen. It is confirmed that the lattice oxygen plays a key role in the catalytic oxidation of toluene over α-MnO2, which follows the Mars–van Krevelen mechanism. Positive hole defects (Mn3+) caused by KOH treatment play an important role in the diffusion of O and enhance the reducibility of manganese oxide. In addition, the enhanced specific surface area, pore volume, and surface acidity are also conducive for the catalytic oxidation of toluene.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...