How coverage influences thermodynamic and kinetic isotope effects for H2/D2 dissociative adsorption on transition metals†
Abstract
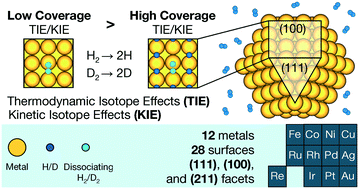
Isotope effects greatly enhance our understanding of chemical reactions in heterogeneous catalysis. Despite their widespread use, there is limited understanding about how realistic reaction conditions, such as the coverage of surface adsorbates, affect them. Here, we study the influence of hydrogen (H) coverage on the thermodynamic and kinetic isotope effects of H2/D2 dissociative adsorption on the close-packed, open, and stepped surfaces of 12 transition metals: Ag, Au, Co, Cu, Fe, Ir, Ni, Re, Pd, Pt, Rh, and Ru, over a catalytically relevant temperature range. Through first-principles density functional theory calculations, we show that increasing coverage has two effects: i) it may change preferred adsorption sites and transition state geometries, and ii) it increases the vibrational frequencies of adsorbed H due to the interactions between H atoms. Empirically, isotope effects decrease in absolute value with increasing coverage for most of our studied systems, indicating a relative shift in stability in favor of the D-substituted minima and transition states. This is likely due to the consistent influence of the latter factor, which affects all structures. Higher temperatures reduce the magnitude of these decreases. Our findings provide insights into the nanoscale mechanisms by which coverage influences isotope effects, which will affect how we interpret experimentally measured isotope effects. They also point towards new applications of isotope effects in catalysis, such as for quantifying adsorbate coverages as well as for elucidating adsorption and active sites on the surfaces of catalysts.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...