Promotion effect of cerium doping on iron–titanium composite oxide catalysts for selective catalytic reduction of NOx with NH3†
Abstract
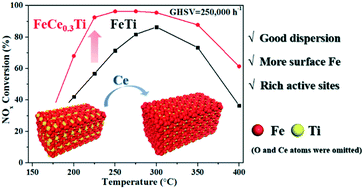
A series of cerium-doped iron–titanium composite oxide catalysts (FeCeaTi, a = 0.1–1.0) prepared by a urea homogeneous precipitation method were investigated for selective catalytic reduction of NOx with NH3 (NH3-SCR). Over all the FeCeaTi samples, a promotion effect for NOx reduction was induced by the introduction of cerium, with the FeCe0.3Ti catalyst exhibiting the best catalytic performance, even at a high GHSV of 250 000 h−1. As indicated by kinetic studies, interestingly, the FeCe0.3Ti catalyst exhibited a higher activation energy for the NH3-SCR process while possessing a pre-exponential factor three orders of magnitude higher than that of FeTi. Going deeper, extensive characterization including N2-physisorption, XRD, Raman, NH3/NOx-TPD, XPS, EPR, and H2-TPR was carried out. XRD and Raman results showed that the introduction of suitable amounts of Ce into the FeTi samples promoted the dispersion of Fe and Ti. Such higher dispersion of these two components increased the capacities for NOx adsorption and activation and weakly bonded ammonia over FeCe0.3Ti, thus promoting the occurrence of the L–H pathway of NH3-SCR at low temperatures. H2-TPR results indicated that the reduction of FeCe0.3Ti occurred at a higher temperature than that of FeTi, which may be a reason for its higher activation energy for NH3-SCR.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...