Mechanistic insight into the organocalcium-mediated nucleophilic alkylation of benzene and further rational design†
Abstract
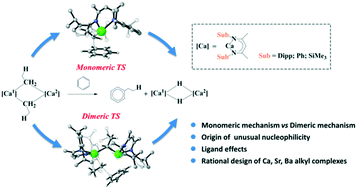
DFT calculations have been performed to study the recently reported unprecedented nucleophilic alkylation of benzene mediated by an organocalcium compound (Science, 2017, 358, 1168). The study reveals that the dimeric reaction mechanism is kinetically more favorable, and the rate-determining step is predicted to be the first nucleophilic attack of benzene by the organocalcium compound. Remarkably, the dimeric mechanism involves the formation of NMR detected dimeric alkyl calcium analogue 4, which agrees well with the experimental observations. EDA-NOCV analysis indicated that the origin of the higher reactivity of alkylcalcium 1 lies in its unusual bonding nature. We further studied the ligand effects and rationally designed the [(SiMe3BDI)CaEt]2 molecule, which shows a higher reactivity towards benzene. In addition, we de novo designed the more reactive heavier alkylstrontium and alkylbarium congeners, which might be targets for experimental synthesis.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...