Role of tungsten modifiers in bimetallic catalysts for enhanced hydrodeoxygenation activity and selectivity†
Abstract
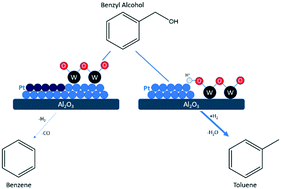
Bimetallic catalysts consisting of a noble metal and an oxophilic modifier have shown promise for upgrading lignin-derived molecules through hydrodeoxygenation (HDO). However, the mechanism of cooperation between the metals is not well understood currently. In this study, we investigated the HDO of gas-phase benzyl alcohol on W-modified Pt/Al2O3 catalysts. Our results show that bimetallic catalysts with an intermediate W loading have enhanced activity and selectivity for HDO compared to a baseline, monometallic Pt/Al2O3 catalyst. Characterization of the active site through CO chemisorption and CO diffuse-reflectance infrared spectroscopy suggests that decarbonylation is suppressed on bimetallic catalysts through site blocking and electronic effects. Based on results from X-ray photoelectron spectroscopy and titration with bases, enhancements in HDO activity were attributed to new active sites in the form of Brønsted acid centers on partially reduced WOx generated through a bifunctional H2 spillover interaction with Pt.
- This article is part of the themed collection: Catalysis Science & Technology 10th Anniversary Symposium


 Please wait while we load your content...
Please wait while we load your content...