A mutually isolated nanodiamond/porous carbon nitride nanosheet hybrid with enriched active sites for promoted catalysis in styrene production†
Abstract
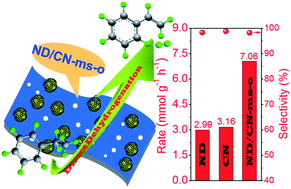
Metal-free carbon-based materials have led to a great breakthrough in energy-saving styrene production via the direct dehydrogenation (DDH) of ethylbenzene in comparison to potassium-promoted iron oxide catalysts that suffer from severe coke formation, drastic deactivation and vast energy consumption. Owing to their unique structure and surface chemistry, nanodiamonds (NDs) have attracted a great deal of attention in heterocatalysis, including the catalytic DDH of ethylbenzene. However, incurable aggregation caused by existing surface forces and chemical bonding forces inevitably causes a deterioration in their catalytic performance due to the lowered accessibility of active sites. Herein, we, for the first time, report a facile two-step molten salt-oxidation approach to fabricate a mutually isolated ND/porous carbon nitride nanosheet hybrid with enriched surface ketonic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O catalytically active sites (ND/CN-ms-o) through de-aggregating NDs and then inserting the well-dispersed NDs into in situ pore-making carbon nitride nanosheets (CNs) in molten salt (MS), followed by oxidation treatment in air. The resulting ND/CN-ms-o catalyst demonstrates 2.4 and 2.3 times higher steady-state styrene rates (7.06 mmol g−1 h−1) towards the direct dehydrogenation of ethylbenzene to styrene compared with pristine NDs (2.99 mmol g−1 h−1) and CNs (3.66 mmol g−1 h−1), respectively. Moreover, this work opens up a new horizon for fabricating other hybrids from dispersion-requiring carbonaceous parents with potential for diverse applications, including catalysis, drug delivery, biosensors, field-emission displays, optoelectronic devices, and chromatographic separation.
O catalytically active sites (ND/CN-ms-o) through de-aggregating NDs and then inserting the well-dispersed NDs into in situ pore-making carbon nitride nanosheets (CNs) in molten salt (MS), followed by oxidation treatment in air. The resulting ND/CN-ms-o catalyst demonstrates 2.4 and 2.3 times higher steady-state styrene rates (7.06 mmol g−1 h−1) towards the direct dehydrogenation of ethylbenzene to styrene compared with pristine NDs (2.99 mmol g−1 h−1) and CNs (3.66 mmol g−1 h−1), respectively. Moreover, this work opens up a new horizon for fabricating other hybrids from dispersion-requiring carbonaceous parents with potential for diverse applications, including catalysis, drug delivery, biosensors, field-emission displays, optoelectronic devices, and chromatographic separation.


 Please wait while we load your content...
Please wait while we load your content...