Self-assembled CoTiO3 nanorods with controllable oxygen vacancies for the efficient photochemical reduction of CO2 to CO†
Abstract
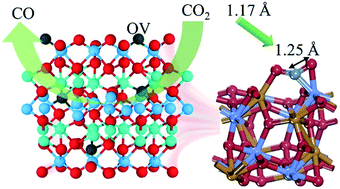
Cobalt titanate (CoTiO3) is a kind of semiconductor material with low catalytic activity, so it is seldom reported. In this work, a simple but effective method was utilized to generate oxygen vacancies on the surface of cobalt titanate. The surface oxygen vacancies were measured by XPS of O 1s and EPR spectroscopy. The presence of oxygen vacancies can not only improve the adsorption ability of CO2, but also change the band structure of CoTiO3, which facilitates the transfer of excited electrons from a photosensitizer to the cocatalyst. Theoretical calculations indicated that the formation of surface oxygen vacancies on CoTiO3 could greatly enhance the binding strength of CO2. Furthermore, the free energy needed to pass the rate-determining step was small during the catalytic process. As a result, the catalytic efficiency of CoTiO3 with oxygen vacancies was nine times that of pristine CoTiO3.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...