Effects of alumina morphology on dry reforming of methane over Ni/Al2O3 catalysts†
Abstract
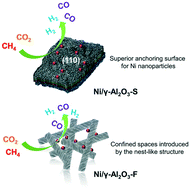
Ni-based catalysts supported on nanosheet (S), nanofiber (F) and particle (P) alumina were successfully designed for the dry (CO2) reforming of methane reaction. The Ni/Al2O3-S catalyst exhibited excellent catalytic activity because the (110) plane of Al2O3-S provides a superior anchoring surface for Ni nanoparticles. During the stability test, the Ni/Al2O3-F catalyst achieved the highest stability with no sign of deactivation, whereas the other catalysts were obviously deactivated under the same reaction conditions. This is because confined spaces were introduced by the nest-like structure of the Al2O3-F support for the confinement of Ni particles. Furthermore, the surface characteristics of the alumina supports and catalysts were analyzed via in situ diffuse reflectance infrared Fourier transform (DRIFT) spectroscopy using CO2 as a probe molecule. In situ DRIFT spectroscopy suggested that the coordinated environment of Al3+ and the surface hydroxyl groups of alumina were altered by the different morphologies. Al2O3-F possessed more basic sites, which readily and stably adsorbed bicarbonate (b-HCO3−) species on the Ni/Al2O3-F catalyst. On the contrary, Ni/Al2O3-S possessed more acidic sites, which adsorbed mono-dentate carbonate (m-CO32−) species, and its chemisorption was less robust and unsteady. The results suggest that the alumina morphology affects the catalytic performance of the dry reforming of methane reaction.


 Please wait while we load your content...
Please wait while we load your content...