The catalytic mechanism of the Au@TiO2−x/ZnO catalyst towards a low-temperature water-gas shift reaction†
Abstract
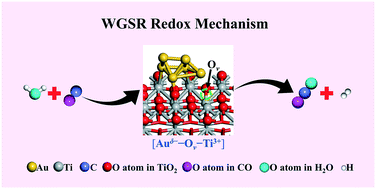
The mechanism of a low-temperature water-gas shift reaction (LT-WGSR) over supported gold catalysts has long been disputed, with the two prevailing ones being redox and associative mechanisms, due to the rather limited direct and reliable evidence for the actual reaction route. Herein, a typical supported gold-based catalyst (Au@TiO2−x/ZnO) was obtained via a structural topotactic transformation from hydrotalcite precursors, exhibiting an extremely high catalytic performance towards LT-WGSR. A series of experimental measurements involving in situ/operando DRIFTS, in situ Pulse-MASS and in situ/operando EXAFS as well as density functional theory (DFT) calculations were carried out to monitor the dynamic evolution of active sites, detect reaction intermediates and identify the reaction route. A new redox reaction route derived from the interfacial synergistic catalysis was verified: one H2O molecule undergoes dissociation at an Auδ−–Ov–Ti3+ (Ov: oxygen vacancy) interfacial active site, accompanied with the production of one H2 molecule and the oxidation of the interfacial structure to Au0–Or–Ti4+ (Or: the interfacial reactive oxygen species). Subsequently, one chemisorbed CO molecule captures Or to produce CO2 with the recovery of Auδ−–Ov–Ti3+. The present understanding and identification of the reaction mechanism of LT-WGSR would offer inspiration for further material exploration and catalyst design in gold-based heterogeneous catalysis.


 Please wait while we load your content...
Please wait while we load your content...