Study of the carbon cycle of a hydrogen supply system over a supported Pt catalyst: methylcyclohexane–toluene–hydrogen cycle†
Abstract
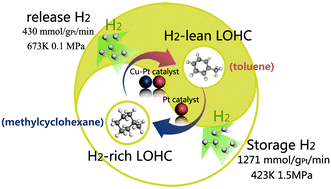
Owing to the importance of the carbon cycle and the universality of carbon loss in sustainable hydrogen supply systems, Pt-based catalysts were designed carefully for a reversible methylcyclohexane–toluene–hydrogen (MTH) cycle. Through adjusting the activity of the Pt species toward C–H bonds to a middle level, liquid organic hydrogen carriers (LOHCs) with higher selectivity were achieved, making the MTH cycle closer to a closed carbon cycle. Specifically, in the hydrogen generation process, we tried to enhance coking resistance as well as inhibit side-reactions such as disproportionation, de-alkylation and hydro-isomerization via Cu doping in Pt/S-1. Experimentally, the selectivities of H2-rich and H2-lean LOHCs in the MTH cycle reached 99.9% with almost coke free during these two processes. Moreover, Pt-based catalysts exhibited excellent activity and durability in catalyzing the MTH cycle. The hydrogen release rate reached 445.3 mmol gPt−1 min−1 with 92.26% methylcyclohexane (MCH) conversion and a hydrogen storage rate of 1271 mmol gPt−1 min−1 was achieved with greater than 99.9% toluene conversion. Finally, through further investigating the geometric configuration and electronic environment of the Cu and Pt species, we found that the Pt was co-located and interacted with the Cu by forming an alloy. Moreover, the unique electron structure of the Cu–Pt alloy facilitated electron transfer from the Pt to the Cu, decreasing the electron density of the Pt. As a consequence, over-dehydrogenation and hydrogenolysis in the MTH cycle were inhibited.


 Please wait while we load your content...
Please wait while we load your content...