Embedding oxygen vacancies at SnO2–CNT surfaces via a microwave polyol strategy towards effective electrocatalytic reduction of carbon-dioxide to formate†
Abstract
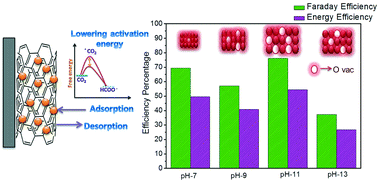
Coupling electrochemical CO2 reduction with renewable energy sources is one of the best ways to minimize the carbon footprint in the atmosphere. Viable technology can be designed with the aid of an electrocatalyst which can be easily scalable and selectively reduces CO2 at lower overpotentials, i.e. ought to be energy efficient. In this context, we utilised the microwave polyol method to synthesis and systematically tune the oxygen vacancies of an SnO2–CNT composite by varying the particle size. The extent of the oxygen vacancy formed was confirmed with the help of Raman, XPS and EPR studies. The oxygen vacancy has the ability to lower the activation barrier for CO2˙− formation. Oxygen vacancy variation has altered the onset potential and the selectivity towards formate production of SnO2–CNT composites. A distinction in the electrocatalytic activity may arise from the difference in CO2 adsorption and intermediate binding strength and desorption. Among all the catalysts, SnO2–CNT pH-11 stands out with the highest faraday and energy efficiency of 76 and 54% towards formate production at −0.77 V vs. RHE, respectively. This catalyst exhibits an excellent stability and produces 16 mM of formate with ∼60% Faraday efficiency after a long term run of 10 hours.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...