Photocatalytic selective aerobic oxidation of amines to nitriles over Ru/γ-Al2O3: the role of the support surface and the strong imine intermediate adsorption†
Abstract
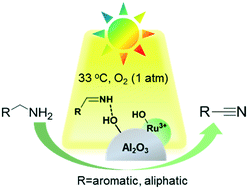
Hydroxyl coordinated ruthenium dispersed on the surface of γ-Al2O3 can be applied to the selective oxidation of amines with light irradiation and an atmospheric pressure of O2 at room temperature. Sunlight is also an effective light source for the selective aerobic oxidation of primary amines to corresponding nitriles. The high photocatalytic activity and selectivity over Ru/γ-Al2O3 originate from the adsorption of amines and imine intermediates on the abundant surface OH groups of the photocatalyst and further formation of Ru–amide species by ligand exchange of adsorbed amines and imine intermediates with adjacent exposed active Ru sites. Light is introduced to the system successfully via the formation of Ru–amide species, which are used as the light absorption sites of the photocatalytic selective oxidation of amines. Primary amines are directly converted to corresponding nitriles via a two-step oxidative dehydrogenation process.


 Please wait while we load your content...
Please wait while we load your content...