Synthesis of a CeO2/Co3O4 catalyst with a remarkable performance for the soot oxidation reaction†
Abstract
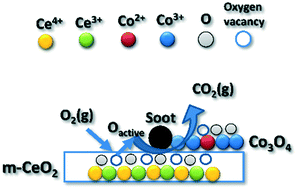
A highly active catalyst based on two metal oxides, CeO2/Co3O4, for the low temperature soot oxidation at low temperatures is reported. This catalyst was produced by depositing cobalt oxide by the method of deposition−precipitation with urea onto CeO2 microstructures obtained by the hydrothermal method using urea and citric acid as precipitating agents. Catalytic experiments were performed by temperature programmed combustion, in a fixed-bed microreactor, under loose and tight contact conditions, using Printex-U (Evonik) as the model soot. The results showed that the CeO2/Co3O4 material presented an enhanced activity for the soot oxidation reaction (50% soot conversion at 287 °C) compared with CeO2 (50% soot conversion at 337 °C) and Co3O4 (50% soot conversion at 360 °C) catalysts separately. The catalytic activity for the CeO2/Co3O4 catalyst is higher than the ones reported for Ce–Co based catalysts, tested under similar conditions, for soot oxidation (reduced by 46 °C the lowest temperature reached for the 50% soot conversion).


 Please wait while we load your content...
Please wait while we load your content...