The stability of Pd/TS-1 and Pd/silicalite-1 for catalytic oxidation of methane – understanding the role of titanium†
Abstract
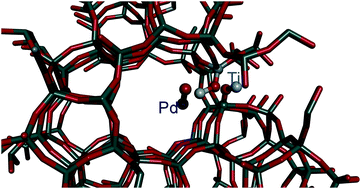
The stability of Pd/TS-1 and Pd/silicalite-1 catalysts was assessed at 400 °C and an approximate relative humidity (RH) of 80% for catalytic combustion of fugitive methane emissions at low concentrations, aiming to understand the role of titanium in the stability of the catalysts. The long-term stability of both catalysts is attributed to both hydrophobicity and low acidity of aluminium-free zeolites. The structure and bonding of Pd deposited on the TS-1 catalyst were fully evaluated using a number of techniques including EXAFS, XPS and TEM. Pd/silicalite-1, in contrast to Pd supported on TS-1, displayed a gradual deactivation over 30 h time-on-stream. This deactivation occurred coincident with the agglomeration of palladium atoms on the surface of this catalyst as well as carbon deposition, whereas this phenomenon was not observed for the Pd/TS-1 material. It is concluded that the hydrophobicity and low acidity is the reason for the high stability and activity of Pd supported on TS-1. In contrast, the dispersion of palladium in Pd/silicalite-1 (and indeed Pd on alumina) decreased remarkably and carbon deposits were formed on the catalyst, contributing to the deactivation. We showed by both, Ti K and Pd K EXAFS analysis an anchoring role of Ti for Pd and confirmed the location of the Pd active in the combustion. It is suggested that the Ti in the T positions of the TS-1 framework forms the sites for palladium particles at an average distance of 2.5 Å and inhibits the sintering of palladium particles. In contrast to acidic supports, we did not observe deactivation by deposition of carbon containing species. Hence, the notable stability of Pd/TS-1 over 1900 h of reaction results in Pd/TS-1 being the most promising catalyst for methane combustion in industrial applications.


 Please wait while we load your content...
Please wait while we load your content...