Understanding the role of surface basic sites of catalysts in CO2 activation in dry reforming of methane: a short review
Abstract
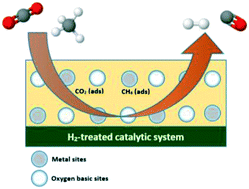
The surface of basic sites in catalysts plays an important role in many catalytic applications, such as dry reforming of methane for generating renewable hydrogen energy. The reaction concept of methane dry reforming is initiated by an acid–base interaction, followed by catalytic cycles. For instance, the reactants of CO2 act as acids toward catalysts, which act as bases. The basic sites of catalysts are generated from a severe pre-treatment temperature by desorbing the surface species. The current consensus on the role of basic sites is to enhance the activation of acidic CO2 on the catalyst support's surface and to inhibit carbon deposition on the catalyst, thus enhancing the catalytic stability. This review elucidated the behaviour of surface basic sites towards CO2 activation in dry reforming of methane. The method for characterising the basicity of catalysts was also reviewed to strategically design catalysts, which could increase the catalytic activity.


 Please wait while we load your content...
Please wait while we load your content...