Design of small molecules targeting RNA structure from sequence†
Abstract
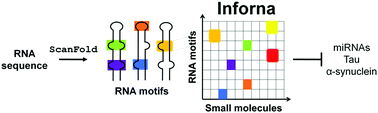
The design and discovery of small molecule medicines has largely been focused on a small number of druggable protein families. A new paradigm is emerging, however, in which small molecules exert a biological effect by interacting with RNA, both to study human disease biology and provide lead therapeutic modalities. Due to this potential for expanding target pipelines and treating a larger number of human diseases, robust platforms for the rational design and optimization of small molecules interacting with RNAs (SMIRNAs) are in high demand. This review highlights three major pillars in this area. First, the transcriptome-wide identification and validation of structured RNA elements, or motifs, within disease-causing RNAs directly from sequence is presented. Second, we provide an overview of high-throughput screening approaches to identify SMIRNAs as well as discuss the lead identification strategy, Inforna, which decodes the three-dimensional (3D) conformation of RNA motifs with small molecule binding partners, directly from sequence. An emphasis is placed on target validation methods to study the causality between modulating the RNA motif in vitro and the phenotypic outcome in cells. Third, emergent modalities that convert occupancy-driven mode of action SMIRNAs into event-driven small molecule chemical probes, such as RNA cleavers and degraders, are presented. Finally, the future of the small molecule RNA therapeutics field is discussed, as well as hurdles to overcome to develop potent and selective RNA-centric chemical probes.
- This article is part of the themed collection: Chemistry Biology Interface Primer


 Please wait while we load your content...
Please wait while we load your content...