C–C and C–X coupling reactions of unactivated alkyl electrophiles using copper catalysis
Abstract
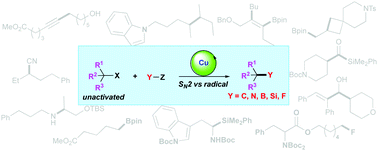
Transition metal-catalysed cross-coupling reactions are widely used for construction of carbon–carbon and carbon–heteroatom bonds. However, compared to aryl or alkenyl electrophiles, the cross-coupling of unactivated alkyl electrophiles containing β hydrogens remains a challenge. Over the past few years, the use of suitable ligands such as bulky phosphines or N-heterocyclic carbenes (NHCs) has enabled reactions of unactivated alkyl electrophiles not only limited to the traditional cross-coupling with Grignard reagents, but also including a diverse range of organic transformations via either SN2 or radical pathways. This review provides a comprehensive overview of the recent development in copper-catalysed C–C, C–N, C–B, C–Si and C–F bond-forming reactions using unactivated alkyl electrophiles.
- This article is part of the themed collections: 2020 Emerging Investigators and Earth Abundant Metals in Catalysis


 Please wait while we load your content...
Please wait while we load your content...