Confronting FCP structure with ultrafast spectroscopy data: evidence for structural variations
Abstract
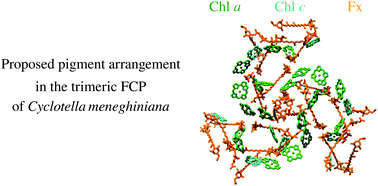
Diatoms are a major group of algae, responsible for a quarter of the global primary production on our planet. Their adaptation to marine environments is ensured by their light-harvesting antenna – the fucoxanthin–chlorophyll protein (FCP) complex, which absorbs strongly in the blue-green spectral region. Although these essential proteins have been the subject of many studies, for a long time their comprehensive description was not possible in the absence of structural data. Last year, the 3D structures of several FCP complexes were revealed. The structure of an FCP dimer was resolved by crystallography for the pennate diatom Phaeodactylum tricornutum [W. Wang et al., Science, 2019, 363, 6427] and the structure of the PSII supercomplex from the centric diatom Chaetoceros gracilis, containing several FCPs, was obtained by electron microscopy [X. Pi et al., Science, 2019, 365, 6452; R. Nagao et al., Nat. Plants, 2019, 5, 890]. In this Perspective article, we evaluate how precisely these structures may account for previously published ultrafast spectroscopy results, describing the excitation energy transfer in the FCP from another centric diatom Cyclotella meneghiniana. Surprisingly, we find that the published FCP structures cannot explain several observations obtained from ultrafast spectroscopy. Using the available structures, and results from electron microscopy, we construct a trimer-based FCP model for Cyclotella meneghiniana, consistent with ultrafast experimental data. As a whole, our observations suggest that the structures from the proteins belonging to the FCP family display larger variations than the equivalent LHC proteins in plants, which may reflect species-specific adaptations or original strategies for adapting to rapidly changing marine environments.
- This article is part of the themed collection: PCCP Perspectives


 Please wait while we load your content...
Please wait while we load your content...