Understanding the luminescence properties of Cu(i) complexes: a quantum chemical perusal†
Abstract
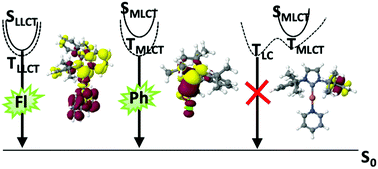
Electronic structures and excited-state properties of Cu(I) complexes with varying coordination numbers have been investigated by means of advanced quantum chemical methods. The computational protocol employs density functional-based methods for geometry optimizations and vibrational analyses including solvent effects through continuum models. Excitation energies, spin–orbit couplings and luminescence properties are evaluated using multireference configuration interaction methods. Rate constants of spin-allowed and spin-forbidden transitions have been determined according to the Fermi golden rule. The computational results for the 4-coordinate (DPEPhos)Cu(PyrTet), the 3-coordinate [IPr–Cu–Py2]+, and the linear CAACMe2–Cu–Cl complexes agree well with experimental absorption and emission wavelengths, intersystem crossing (ISC) time constants, and radiative lifetimes in liquid solution. Spectral shifts on the ligand-to-ligand charge transfer (LLCT) and metal-to-ligand charge transfer (MLCT) transitions caused by the polarity of the environment are well represented by the continuum models whereas the shifts caused by pseudo-Jahn–Teller distortions in the MLCT states are too pronounced in comparison to solid-state data. Systematic variation of the ligands in linear Cu(I) carbene complexes shows that only those complexes with S1 and T1 states of LLCT character possess sufficiently small singlet–triplet energy gaps ΔEST to enable thermally activated delayed fluorescence (TADF). Complexes whose S1 and T1 wavefunctions are dominated by MLCT excitations tend to emit phosphorescence instead. Unlike the situation in metal-free TADF emitters, the presence of low-lying locally excited triplet states does not promote ISC. These states rather hold the danger of trapping the excitation with nonradiative deactivation being the major deactivation channel.
- This article is part of the themed collections: Quantum Theory: The Challenge of Transition Metal Complexes and 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...