A novel amalgamation of deep eutectic solvents and crowders as biocompatible solvent media for enhanced structural and thermal stability of bovine serum albumin†
Abstract
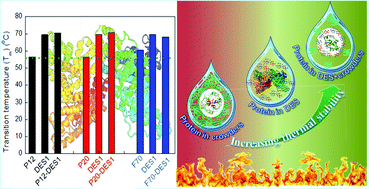
Proteins have immense untapped potential in numerous industries as a green catalyst. Thus, there is an emergent need to find a suitable co-solvent which is biocompatible with proteins and environmentally safe. In this context, the present study investigates the effect of a novel solvent medium designed by an amalgamation of macromolecular crowders and deep eutectic solvents (DESs) on bovine serum albumin (BSA). It was discovered that the presence of crowding agents such as polyethylene glycol (PEG) 12 kDa (P12), 20 kDa (P20), and Ficoll 70 kDa (F70) marginally destabilizes the conformational stability of BSA. While the thermal stability of BSA was dramatically enhanced in the presence of choline chloride (ChCl)-based deep eutectic solvents (DESs) namely ChCl-urea (DES1) and ChCl-glycerol (DES2), the order of stability was (DES1) > (DES2). It was interesting to note that DES1 possessing urea as a hydrogen bond donor leads to the exceptional thermal stability of the BSA structure. Taking a cue from this, an innovative crowded DES medium was prepared by mixing a synthetic crowder and DES1 in optimum concentration. Fascinatingly, the combination of DES1 with PEG delivers promising results, as they elevate the thermal stability of BSA by approximately 16 °C. Thus, crowded DES medium confers structural compactness to the BSA. Altogether, this work reports for the first time the potential of DESs to attenuate the adverse effects of macromolecular crowding on protein stability.


 Please wait while we load your content...
Please wait while we load your content...