Specific and non-specific interactions between metal cations and zwitterionic alanine tripeptide in saline solutions reported by the symmetric carboxylate stretching and amide-II vibrations†
Abstract
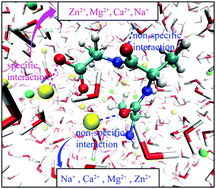
The “specific” interaction between metal cations (Na+, Ca2+, Mg2+, and Zn2+) and the charged COO− group, and the “non-specific” interaction between these cations and the peptide backbone of a zwitterionic trialanine (Ala3) in aqueous solutions were examined in detail, using linear infrared (IR) absorptions of the COO− symmetric stretching and the amide-II (mainly the C–N stretching) modes as IR probes. Different IR spectral changes in peak positions and intensities of the two IR probes clearly demonstrate their sensitivities to nearby cation distributions in distance and population. Quantum chemistry calculations and molecular dynamics simulations were used to describe the cation–peptide interaction picture. These combined results suggest that Na+ and Ca2+ tend to bind to the COO− group in the bidentate form, while Mg2+ and Zn2+ tend to bind to the COO− group in the pseudo-bridging form. The results also show that while all three divalent cations indirectly interact with the peptide backbone with large population, Ca2+ and Mg2+ can be sometimes distributed very close to the backbone. Such a non-specific cation interaction can be moderately sensed by the C–N stretching of the amide-II mode when cations approach the polar amide C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group, and is also influenced by the NH3+ charge group located at the N-terminus. The results suggest that the experimentally observed complication of the Hofmeister cation series shall be understood as a combined specific and non-specific cation–peptide interactions.
O group, and is also influenced by the NH3+ charge group located at the N-terminus. The results suggest that the experimentally observed complication of the Hofmeister cation series shall be understood as a combined specific and non-specific cation–peptide interactions.


 Please wait while we load your content...
Please wait while we load your content...