Molecular recognition at the air–water interface: nanoarchitectonic design and physicochemical understanding
Abstract
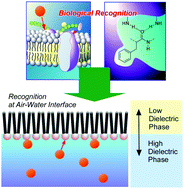
Although molecular recognition at the air–water interface has been researched for over 30 years, investigations on its fundamental aspects are still active research targets in current science. In this perspective article, developments and future possibilities of molecular recognition at the air–water interface from pioneering research efforts to current examples are overviewed especially from the physico-chemical viewpoints. Significant enhancements of binding constants for molecular recognition are actually observed at the air–water interface although molecular interactions such as hydrogen bonding are usually suppressed in aqueous media. Recent advanced analytical strategies for direct characterization of interfacial molecules also confirmed the promoted formation of hydrogen bonding at the air–water interfaces. Traditional quantum chemical approaches indicate that modulation of electronic distributions through effects from low-dielectric phases would be the origin of enhanced molecular interactions at the air–water interface. Further theoretical considerations suggest that unusual potential changes for enhanced molecular interactions are available only within a limited range from the interface. These results would be related with molecular recognition in biomolecular systems that is similarly supported by promoted molecular interactions in interfacial environments such as cell membranes, surfaces of protein interiors, and macromolecular interfaces.
- This article is part of the themed collections: PCCP Perspectives and 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...