Silica based ionogels: interface effects with aprotic and protic ionic liquids with lithium†
Abstract
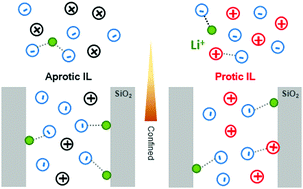
In the frame of the development of solid ionogel electrolytes with enhanced ion transport properties, this paper investigates ionogel systems constituted by ∼80 wt% of ionic liquids (ILs) confined in meso-/macroporous silica monolith materials. The anion–cation coordination for two closely related ILs, either aprotic (AIL) butylmethylpyrrolidinium or protic (PIL) butylpyrrolidinium, both with bis(trifluoromethylsulfonyl)imide (TFSI) anions, with and without lithium cations, is studied in depth. The ILs are confined within silica with well-defined mesoporosities (8 to 16 nm). The effects of this confinement, onto melting points, onto conductivity followed by impedance spectroscopy, and onto lithium–TFSI coordination followed by Raman spectroscopy, are presented. Opposite effects have been observed on the melting temperature: it increased for the AIL (+2 °C) upon confinement, while it decreased for the PIL (−2 °C). With lithium, the confinement led to an increase of the melting temperature (+1 °C) for the PIL and AIL. Regarding ionic conductivities, a relative maximum was observed at 40 °C for a mesopore diameter of 10 nm for the AIL with 0.5 M lithium, while it was not clearly visible for the PIL. These differences are discussed in view of the charge balance at the interface between silanols and ILs: the presence of a PIL, contrary to an AIL, is expected to modify the acidity of the silica. Raman data showed that the coordination number of lithium by TFSI is reduced upon AIL confinement, although this was not observed for PILs. At last, this work highlights the impact of the acidity of a PIL on the chemistry occurring at the interface of the host network within ionogels.


 Please wait while we load your content...
Please wait while we load your content...