Conformational topography of tris(2-methylbutyl) phosphate and the influence of methyl branching at the non-hyperconjugative carbon on the conformational landscape: insights from matrix isolation infrared spectroscopy and DFT computations†
Abstract
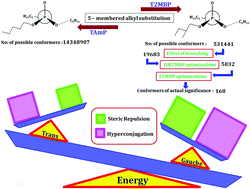
The branching of a methyl group in a linear chain has a profound influence on the conformational morphology as it wields a strong control in reducing a large number of conformations. To unravel the effect of branching on the second non-hyperconjugative carbon atom on the conformational landscape, the conformations of tris(2-methylbutyl)phosphate (T2MBP) were studied using Density Functional Theory (DFT) computations and matrix isolation infrared spectroscopy. Experimentally, T2MBP along with N2/Ar/Kr/Xe gases was effusively expanded and deposited at a low temperature of 12 K, which was subsequently probed using infrared spectroscopy. The computations of all the conformations were accomplished using the B3LYP level of theory with the 6-311++G(d,p) basis set. A dimethyl(2-methylbutyl) phosphate (DM2MBP) prototype, a molecule containing a single 2-methylbutyl moiety, was examined for its conformations. Computations predicted 18 and 9 conformations each for the ‘gauche’ and ‘trans’ families, respectively, in which the third branched carbon completely influences the orientation of the fourth carbon, which simplifies the conformational problem of DM2MBP. Of the 18 and 9 bunches each in the ‘gauche’ and ‘trans’ families, only 7 and 3 conformations, respectively, became energetically important, which when extrapolated to T2MBP resulted in 343 and 147 conformational possibilities. The factor of degeneracy further reduced these numbers and a total of 168 conformations effectively contribute to the conformational composition of T2MBP in the gas phase. The role of stereo electronic and steric factors prevalent in the conformational clusters of T2MBP was unravelled respectively using natural bond orbital and non-covalent interaction analyses.


 Please wait while we load your content...
Please wait while we load your content...