A theoretical study of adsorption on iron sulfides towards nanoparticle modeling†
Abstract
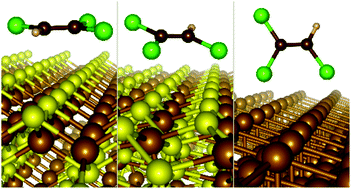
Surface modification of zero-valent iron (nZVI) nanoparticles, which are frequently used in the removal of chlorinated hydrocarbons from contaminated groundwater, can increase their surface stability without significant loss of reactivity. Sulfidation is a process during which thin iron sulfide phases are formed on nZVI particles. In this work, the adsorption capability of two iron sulfide minerals (mackinawite and pyrite) and ZVI with respect to two small polar molecules (H2O and H2S) and trichloroethylene (TCE) was modeled by using the quantum mechanics (QM) approach. High-level QM methods used on cluster models helped in benchmarking and validation of density functional theory methods used on periodic slab models of the (001) surfaces of iron sulfides and the (111) surface of ZVI. This careful computational treatment was necessary for achieving reliable results because modeled iron containing compounds represent computationally demanding systems. The results showed that adsorption was strongly affected by surface topology, accessibility of surface sites, and the shape of adsorbed molecular species. The mackinawite surface is practically hydrophobic having weak interactions with polar molecules (about −5/−6 kcal mol−1), in contrast to the surfaces of pyrite and ZVI (adsorption energies are about three times larger). On the other hand, the adsorption of weakly polar planar TCE molecule is relatively strong and similar for all three surfaces (in the range of −11 to −17 kcal mol−1). Moreover, it was shown that the dominant component of the adsorption energy of TCE had originated from dispersion interactions, which were less important for small polar molecules.


 Please wait while we load your content...
Please wait while we load your content...