Theoretical study on the interaction of iodide electrolyte/organic dye with the TiO2 surface in dye-sensitized solar cells†
Abstract
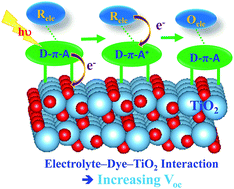
The iodide/triiodide interaction with the dye on a semiconductor surface plays a significant role in understanding the dye-sensitized solar cells (DSSCs) mechanism and improving its efficiency. In the present study, density functional theory (DFT) calculations were used to determine the interaction between the complexed iodide redox couple with dye/TiO2 for the relevance of DSSCs. Three new metal-free organic dyes noted as D1Y, D2Y and D3Y, featured with D–π–A configuration were designed by varying functional groups on the donor moiety. We analyzed the structural and electronic properties of these dyes when standing alone and being adsorbed on the oxide surface with the iodide electrolyte. Of the designed dyes, the incorporation of a strong donor unit in D1Y and D2Y sensitizers in conjunction with iodide electrolytes on the TiO2 surface provides better adsorption and electronic properties in comparison to those from the dye alone on the TiO2 surface. Analysis of density of states (DOS) indicates that the introduction of a strong electron-donating group into the organic dye, mainly D1Y and D2Y with an iodide electrolyte on the surface remarkably upshifts the Fermi energy, thereby improving the efficiency of the DSSCs by an increase of the open-circuit voltage (Voc). The present finding constitutes the basis for achieving a deeper understanding of the intrinsic interaction taking place at the electrolyte/dye/TiO2 interface and provides us with directions for the design of efficient dyes and redox electrolytes for improving DSSCs.


 Please wait while we load your content...
Please wait while we load your content...