Passivation and activation of La0.6Sr0.4FeO3 thin film electrodes†
Abstract
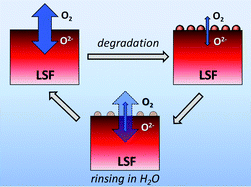
The oxygen exchange activity of thin dense La0.6Sr0.4FeO3 electrodes prepared by pulsed laser deposition was investigated by electrochemical impedance spectroscopy and electrical conductivity relaxation below 600 °C. The value of the surface exchange coefficient (kchem) measured at 491 °C decreased from an initial 4.4 × 10−6 cm s−1 to 1.7 × 10−7 cm s−1 after aging for 7 days. The rapid decrease in the oxygen exchange rate was accompanied by the increase in the surface concentration of ‘non-lattice strontium’ detected by X-ray photoelectron spectroscopy. Subsequent electrochemical tests over 40 days showed that the electrode performance could be recovered by rinsing the passivated electrode in deionized water. Repeated treatments eventually also led to improved stability of electrochemical performance.


 Please wait while we load your content...
Please wait while we load your content...