Effects of catalyst surfaces on adsorption revealed by atomic force microscope force spectroscopy: photocatalytic degradation of diuron over zinc oxide†
Abstract
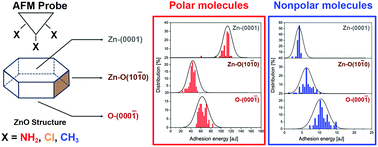
Controlling adsorption of a heterogeneous catalyst requires a detailed understanding of the interactions between reactant molecules and the catalyst surface. Various characteristics relevant to adsorption have been theoretically predicted but have yet to be experimentally quantified. Here, we explore a model reaction based on diuron [3-(3,4-dichlorophenyl)-1,1-dimethylurea] photo-degradation over a ZnO particle catalyst. We used atomic force microscope (AFM)-based force spectroscopy under ambient conditions to investigate interactions between individual functional groups of diuron (NH2, Cl, and CH3) and surfaces of ZnO particles (polar Zn and O-terminated, and nonpolar Zn–O terminated). We were able to distinguish and identify the two polar surfaces of conventional ZnO particles and the nonpolar surface of ZnO nanorods based on force–distance curves of functionalized probe/surface pairs. We posit that the reaction involved physisorption and could be described in terms of Hamaker constants. These constants had an order-of-magnitude difference among the probe/surface interacting pairs based on polarity. Hence, we confirmed that van der Waals interactions determined the adsorption behavior. We interpreted the electronic distribution models of the probe-modifying molecules. The functional group configurations inferred the diuron adsorption configurations during contact with each ZnO facet. The adsorption affected characteristics of the reaction intermediates and the rate of degradation.


 Please wait while we load your content...
Please wait while we load your content...