Insight into the structure-antibacterial activity of amino cation-based and acetate anion-based ionic liquids from computational interactions with the POPC phospholipid bilayer†
Abstract
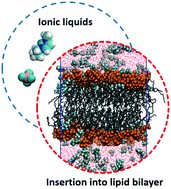
Investigations relevant to ionic liquids (ILs) as antibacterial agents have drawn considerable attention. However, the high cost and potential toxicity of ILs have severely limited their extensive applications, which has motivated researchers to design inexpensive and health-benign ILs. In this work, the interactions between the hydrated zwitterionic phospholipid (POPC) bilayer and a series of hypothetical amino cation-based and acetate anion-based ILs with different counterparts were investigated using molecular dynamics (MD) simulations to predict their antibacterial abilities. The cations of the ILs were found to insert into the lipid bilayer spontaneously, especially amino cations. Reorientation of the inserted imidazolium-based cations was observed, while the inserted amino cations showed no obvious reorientation phenomena, probably because of the strong charge interactions between the positive NH3 groups of the amino cation and the negative PO4 groups of the lipid bilayer. Due to their strong affinity with water, acetate-based anions disperse better in water solution, which weakens the insertion of the cations into the lipid bilayer to some extent. The structure and dynamic properties of the lipid bilayer, such as electrostatic potential, local ordering, area per lipid, volume per lipid, bilayer thickness, and lateral diffusion, are significantly influenced by the insertion of the cations, which results in disorder of the lipid bilayer and further disruption of the activity of the cell membrane. The insights into the relationship between the structures of ILs and their antibacterial activity in this work will provide a good reference for the screening and design of less expensive, safer, and greener IL candidates as antibacterial agents.


 Please wait while we load your content...
Please wait while we load your content...