Understanding the role of ionic flux on the polarity of the exposed surfaces of ZnO†
Abstract
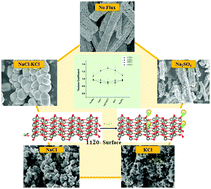
The role of ionic flux in controlling the polarity of the surfaces of ZnO was evaluated, both experimentally and theoretically. Zinc oxide was synthesized by controlled decomposition of zinc oxalate nanorods in the presence of ionic flux. The degree of preferred orientation for a specific plane, for the ZnO structures, was observed by calculating the texture coefficient. The presence of flux (NaCl, KCl, a mixture of NaCl–KCl and Na2SO4) during decomposition of the oxalate precursor led to the preferential growth of (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) planes. The value of texture coefficient was found to be high for the (11
0) planes. The value of texture coefficient was found to be high for the (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) plane when the decomposition was carried out in the presence of a mixture of NaCl and KCl when compared to their counterparts. A decrease in the value of texture coefficient for the (11
0) plane when the decomposition was carried out in the presence of a mixture of NaCl and KCl when compared to their counterparts. A decrease in the value of texture coefficient for the (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) plane was observed when Na2SO4 was used as a flux, which was similar to the value obtained for ZnO synthesized in the absence of flux. The observations from the analysis of texture coefficient were correlated with the photocatalytic degradation of rhodamine B dye, by making use of the fact that the nature of exposed surfaces influences the catalytic activity of a material. On-site Coulomb correlation corrected density functional theory (DFT + U)-based computational studies were performed to get theoretical insight into the role of the ionic flux in surface reconstructions. The surface energies for different ZnO surfaces were computed in the presence and absence of the ionic flux. It was revealed that the pristine (10
0) plane was observed when Na2SO4 was used as a flux, which was similar to the value obtained for ZnO synthesized in the absence of flux. The observations from the analysis of texture coefficient were correlated with the photocatalytic degradation of rhodamine B dye, by making use of the fact that the nature of exposed surfaces influences the catalytic activity of a material. On-site Coulomb correlation corrected density functional theory (DFT + U)-based computational studies were performed to get theoretical insight into the role of the ionic flux in surface reconstructions. The surface energies for different ZnO surfaces were computed in the presence and absence of the ionic flux. It was revealed that the pristine (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surface is more stable compared to pristine (11
0) surface is more stable compared to pristine (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) by 0.04 J m−2 (surface energy), however the scenario changes in the presence of the ionic flux and (11
0) by 0.04 J m−2 (surface energy), however the scenario changes in the presence of the ionic flux and (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) becomes more stable by 0.03 J m−2. This indeed corroborated with our experimental observations and explained the fundamental role of ionic flux on the polarity of exposed surfaces of ZnO.
0) becomes more stable by 0.03 J m−2. This indeed corroborated with our experimental observations and explained the fundamental role of ionic flux on the polarity of exposed surfaces of ZnO.


 Please wait while we load your content...
Please wait while we load your content...