Low-temperature decomposition and segregation on a surface in carbide-containing solid solutions of the zirconium–niobium–carbon system and in related ternary systems†
Abstract
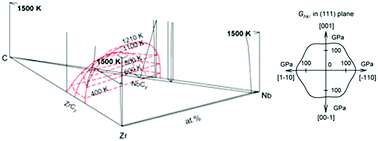
The crystal structure and microstructure of pseudobinary (ZrC0.96)1−x(NbC0.97)x carbide solid solutions has been studied. It was found that monocrystalline grains of zirconium carbide are spontaneously isolated on the surface of diluted solid solutions of the pseudobinary system ZrCy–NbCy′ containing less than 2.0 mol% of zirconium carbide. It is shown that the appearance of zirconium carbide is a consequence of the solid-phase decomposition of these solid solutions and anisotropy of elastic properties of monocrystalline ZrC carbide grains. The model of subregular solutions was used in the temperature interval from 300 to 3900 K to calculate and plot an equilibrium phase diagram of the ternary Zr–Nb–C system. It is shown that at temperatures above 1210 K in the Zr–Nb–C ternary system, nonstoichiometric carbides ZrCy and NbCy′ have unlimited mutual solubility and form a continuous series of (ZrCy)1−x(NbCy′)x solid solutions with 0.6 ≤ y ≤ 0.98, 0.7 ≤ y′ ≤ 1.0, and 0 ≤ x ≤ 1.0. At temperatures below 1200 K, under equilibrium conditions, a discontinuity of the miscibility of the solid solutions ZrCy–NbCy′ is observed and there appears a region of solid phase decomposition. The anisotropy of elastic properties of monocrystalline grains of ZrC was considered. It is predicted that solid-phase decomposition and surface segregation can be observed in such related carbide systems as HfCy–NbCy′, HfCy–TaCy′, ZrCy–TaCy′, VCy–TaCy′ and VCy–NbCy′.


 Please wait while we load your content...
Please wait while we load your content...