Exciton energy transfer reveals spectral signatures of excited states in clusters†
Abstract
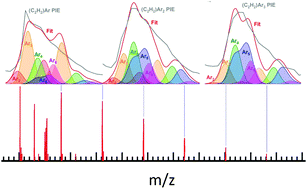
Electronic excitation and concomitant energy transfer leading to Penning ionization in argon–acetylene clusters generated in a supersonic expansion are investigated with synchrotron-based photoionization mass spectrometry and electronic structure calculations. Spectral features in the photoionization efficiency of the mixed argon–acetylene clusters reveal a blue shift from the 2P1/2 and 2P3/2 excited states of atomic argon. Analysis of this feature suggests that excited states of argon clusters transfer energy to acetylene, resulting in its ionization and successive evaporation of argon. Theoretically calculated Arn (n = 2–6) cluster spectra are in excellent agreement with experimental observations, and provide insight into the structure and ionization dynamics of the clusters. A comparison between argon–acetylene and argon–water clusters reveals that argon solvates water better, allowing for higher-order excitons and Rydberg states to be populated. These results are explained by theoretical calculations of respective binding energies and structures.


 Please wait while we load your content...
Please wait while we load your content...