Surface functionalization: an efficient alternative for promoting the catalytic activity of closed shell gold clusters†‡
Abstract
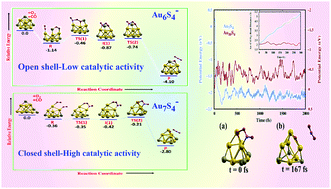
Surface functionalization through adsorption of ligands or non-metal atoms is considered to be an interesting and viable approach for tuning the physicochemical properties of gold clusters. Highly stable and magic numbered electronic configurations of thiolate protected gold clusters such as Au25(SR)18, Au38(SR)24etc. with intriguing properties are the direct manifestation of the rich chemistry of the Au–S interface. The present investigation discerns the CO oxidation activity of structurally well characterized sulphur functionalized gold cluster anions AumS4−, m = 6–10. To establish an in-depth understanding, their activities are analyzed and compared with the corresponding pristine gold clusters. It is seen that sulphur functionalization irrespective of a closed or open shell nature leads to a significant decrease in the O2 adsorption energies on the anionic gold clusters. However, in sharp contrast to O2 adsorption, surface functionalization gives rise to multifarious catalytic behavior in AumS4− clusters with catalytic activity ranging from low (for Au6S4−, Au8S4−) to moderate (for Au9S4−, Au10S4−) to very high (for Au7S4−) for CO oxidation. It is interesting to note that the closed shell Au7S4− and Au9S4− clusters with poor O2 adsorption show remarkably low activation barriers and enhanced catalytic activity as compared to the open shell AumS4− clusters with an odd number of electrons. In particular, in the case of Au7S4− the lowest activation energy barriers of 0.01 and 0.21 eV are obtained, making the CO oxidation reaction facile. Moreover, ab initio molecular dynamics are performed to confirm the enhanced catalytic behaviour of Au7S4− and its dynamical stability during the desorption of CO2 molecule from its surface.


 Please wait while we load your content...
Please wait while we load your content...