Characterization of three phases of liquid carbon by tight-binding molecular dynamics simulations
Abstract
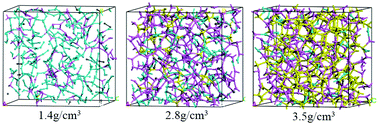
We have performed systematic molecular dynamics simulations to study the structures of liquid carbon at 5000 K with the weight density ranging from 1.4 to 3.5 g cm−3, using a three-center tight-binding potential of carbon. The simulation results show that the bonding characteristics of the liquid changes predominately from twofold to threefold, and then to fourfold coordination as the density increases. Signals of two structural changes at the densities of about 1.9 and 3.0 g cm−3 respectively are revealed by the slope changes in the density dependence of structural, electronic and dynamical properties. Our simulation results suggest that there are three distinct liquid carbon phases in this density range. However, further thermodynamics calculations, e.g., free energy calculations, would be required to clarify the possible liquid–liquid transitions.


 Please wait while we load your content...
Please wait while we load your content...