Highly efficient energy transfer from a water soluble zinc silver indium sulphide quantum dot to organic J-aggregates†
Abstract
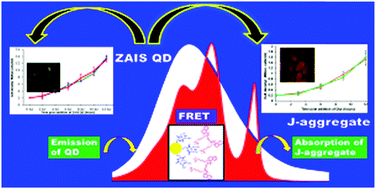
The present work has been carried out with the aim to design and develop an efficient light harvesting inorganic–organic hybrid nanoscale material by employing a less toxic, environment friendly inorganic substance and also to understand the mechanism of inter-particle electronic interaction between the inorganic and organic components of the nanomaterial. Specifically, the inorganic–organic hybrid associate has been made by integrating water soluble semiconductor (zinc–silver–indium–sulfide (ZAIS)) QDs and organic J-aggregates of a cyanine dye (S2165). The fabrication of the present nano-hybrid system has been achieved via electrostatically driven self-assembly of organic dyes over ZAIS QDs. The interaction between QD and J-aggregates has been investigated by using steady state and time resolved fluorescence measurements. Zeta potential measurements have also been performed to understand the role of electrostatic interaction and thermodynamic feasibility of the association process. The investigations have revealed that the energy transfer (ET) process between QD and J-aggregates was mediated through a dipole–dipole mechanism. Interestingly, data analysis based on Förster theory has further revealed that the ET from QD to J-aggregates is very high, indicating efficient electronic coupling between the inorganic QD and the organic J-aggregates. Zeta potential measurements and thermodynamic calculations have demonstrated that the interaction between QD and organic dye is electrostatically driven and the association of organic dyes over QDs is thermodynamically feasible. The outcome of the present study is expected to be helpful in designing efficient nanoscale light harvesting devices. Additionally, fluorescence microscopy and toxicity studies on the QDs have also shown their suitability for biological applications.


 Please wait while we load your content...
Please wait while we load your content...