Highly-excited state properties of cumulenone chlorides in the vacuum-ultraviolet†
Abstract
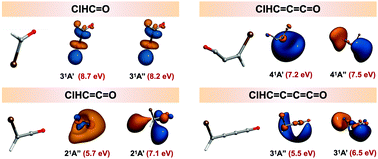
Recent observations of chloromethane in interstellar environments suggest that other organohalogens, which are known to be critically important in Earth's atmosphere, may also be of significance beyond our own terrestrial veil. This raises the question of how such molecules behave under extreme conditions such as when exposed to vacuum ultraviolet (VUV) radiation. VUV photons promote molecules to highly excited states that fragment in non-statistical patterns controlled by the initial femtosecond dynamics. A detailed understanding of VUV-driven photochemistry in complex organic molecules that consist of more than one functional group is a particularly challenging task. This quantum chemical analysis reports the electronic states and ionization potentials up to the VUV range (6–11 eV) of the chlorine-substituted cumulenone series molecules. The valence and Rydberg properties of lone-pair terminated, π-conjugated systems are explored for their potential resonance with lone pairs from elsewhere in the system. The carbon chain elongation within the family ClHCnO, where n = 1–4, influences the electronic excitations, associated wavefunctions, and ionization potentials of the molecules. The predicted geometries and ionization potentials are in good agreement with the available experimental photoelectron spectra for formyl chloride and chloroketene, n = 1–2. Furthermore, comparison between the regular cumulenone species and the corresponding chlorinated derivatives exhibit similar behaviors especially for n = 3, where the allene backbone in propadienone chloride is severely bent. Most notably for the excited states is that the Rydberg character becomes more dominant as the energy increases, with some retaining valence characters.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...