Effect of zeolite morphology on charge separated states: ZSM-5-type nanocrystals, nanosheets and nanosponges†
Abstract
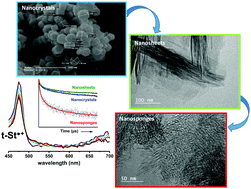
In the present work, we investigate the electron transfer occurring in the porous void of three MFI-type zeolite (ZSM-5) nanomaterials (nanocrystals, nanosheets and nanosponges) after adsorption and photoexcitation of t-stilbene (t-St). ZSM-5 nanosheets are constituted of lamellar stacking of several nanosheets (20–40 nm) where each nanosheet has a thickness of 2 nm. Nanosponges are composed of ZSM-5 nanocrystals (2–3 nm) separated by mesoporous holes of 5.8 nm facilitating the synthesis of hierachical materials. While the nanosheets show microporosity similar to that observed for the ZSM-5 nanocrystals, the absorption isotherms of the nanosponges show the existence of secondary micropores. After photoirradiation of t-St, UV-vis absorption spectroscopy shows the formation of charge separated states (radical cation and charge transfer complex) in the nanocrystals and in the nanosheets whereas no ionized species is detected in the nanosponges. The radical cation (RC) is stabilized in the nanosheets while it evolves very rapidly towards a Charge Transfer Complex (CTC) in the nanocrocrystals. The particular morphology of the nanosheets and nanosponges is put forward to explain this result since all host materials are of the MFI-type. To investigate ultra-short phenomena in the three nanomaterials, the UV-vis transient spectra were recorded between 2 and 450 μs after photoexcitation by nanosecond laser pulses. In the nanocrystals and nanosheets only the RC is detected whereas CTC formation is not observed. Photoexcitation of t-St in the nanosponges also leads to the formation of a RC but it recombines completely within 70 μs. This suggests the preferential location of t-St in the secondary micropores with pores larger than the micropores of the MFI-type framework and possibly in the mesopores of the nanosponges.


 Please wait while we load your content...
Please wait while we load your content...