Salt-induced LCST-type thermal gelation of methylcellulose: quantifying non-specific interactions via fluctuation theory†
Abstract
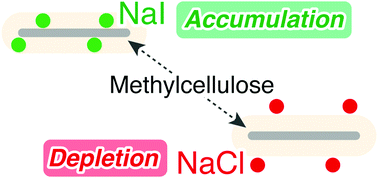
What drives the phase separation of water-soluble polymers in the presence of electrolytes was quantified on a molecular scale via statistical thermodynamic fluctuation theory. Quantifying polymer–water and polymer–salt interactions enabled us to identify the dominant interaction for phase separation. As a model system, the lower critical solution temperature (LCST) type thermal gelation of methylcellulose (MC) in aqueous salt solutions was chosen. The Kirkwood–Buff integrals for intermolecular interactions, calculated from the published calorimetric and volumetric data, showed that (1) the accumulation of salts around MC molecules (favourable interaction between salts and MC) inhibits thermal gelation and the depletion of salts from MC (unfavourable interaction between salts and MC) promotes the gelation, and (2) this salt–MC interaction is the dominant factor (50–100 times stronger than the water–MC interaction). This insight from the fluctuation theory is at odds with the age-old consensus regarding the driving force of thermal gelation: water structure change in the presence of salts induces the promotion or inhibition of thermal gelation. However, our conclusion is founded upon the ability of the fluctuation theory to quantify water–MC and salt–MC interaction independently via the Kirkwood–Buff integrals. Flory–Huggins (FH) theory, on the contrary, could not separate these two interactions owing to the lack of a thermodynamic degree of freedom because the lattice solution is assumed to be fully packed. In addition, the dominant contribution from salt depletion poses difficulty for the χ parameter, which is essentially the difference of contact energies. Our approach, requiring calorimetric and volumetric data alone as input, provides a simple and versatile method towards elucidating the effect of cosolvents on biopolymer phase separation of physiological importance.


 Please wait while we load your content...
Please wait while we load your content...