The effect of introducing an ether group into an imidazolium-based ionic liquid in binary mixtures with DMSO†
Abstract
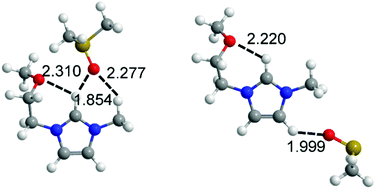
Ether-functionalized ionic liquids (ILs) have successfully been employed in diverse applications, but their interactions with other solvents are not understood well. In this work, mixtures of 1-methoxyethyl-3-methylimidazolium bis(fluorosulfonyl)imide (EOMIMFSI) and dimethylsulfoxide (DMSO) are studied in terms of their solution structure and hydrogen bonding interactions. The corresponding alkyl-substituted IL 1-ethyl-3-methylimidazolium bis(fluorosulfonyl)imide (EMIMFSI) is analyzed for comparison. A combination of FTIR spectroscopy, excess spectroscopy, and quantum chemical calculations is employed for this purpose. The datasets allow drawing a number of conclusions as follows: (1) the ether group forms intramolecular hydrogen bonds that compete with anions and DMSO; hence, introducing ether groups into the imidazolium-based IL leads to the weakening of hydrogen bonds in the mixtures. (2) With the help of excess spectra and quantum chemical calculations, some complexes such as ion clusters, ion pairs, and individual ions were identified and assigned in the two systems. The solution structures at different concentrations were examined by analyzing the excess spectra of ν(C2–H) and ν(C–D) in the two IL–DMSO-d6 systems. (3) The introduced ether groups result in changes of the main interaction sites, which were found to be concentration-dependent. In the EOMIMFSI–DMSO system, when isolated ions are the main existing form, the C2–Hs are still the main sites interacting with DMSO. However, when ion pairs or larger ion clusters are the main existing species, the C4–Hs are the main sites interacting with DMSO.


 Please wait while we load your content...
Please wait while we load your content...