Influence of surface hydrophilicity and hydration on the rotational relaxation of supercooled water on graphene oxide surfaces†
Abstract
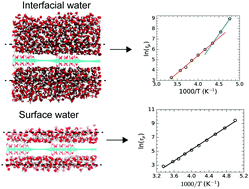
Hydration or interfacial water present in biomolecules and inorganic solids has been shown to exhibit a dynamical transition upon supercooling. However, understanding the extent of the underlying surface hydrophilicity as well as the local distribution of hydrophilic/hydrophobic patches on the dynamical transition is unexplored. Here, we use molecular dynamics simulations with a TIP4P/2005 water model to study the translational and rotational relaxation dynamics of interfacial water on graphene surfaces. The purpose of this study is to investigate the influence of both the surface chemistry and the extent of hydration on the rotational transitions of interfacial water on graphene oxide (GO) surfaces in the deeply supercooled region. We have considered three graphene-based surfaces: a GO surface with equal proportions of oxidized and pristine graphene regions in a striped topology, a fully oxidized surface and a pristine graphene surface. The dipole relaxation time of interfacial water (high hydration) shows a strong-to-strong transition, strong nature, and strong-to-strong transition on these surfaces, respectively, in the temperature range of 210–298 K. In contrast, bulk water shows a fragile-to-strong rotational transition upon supercooling. In all these cases at high hydration, interfacial water co-exists with a thick water film with bulk-like properties. To investigate the influence of bulk water on dynamical transitions, we simulated a low hydration regime where only bound water (surface water) is present on the GO surfaces and found that the rotational relaxation of surface water on both the GO and fully oxidized surfaces shows a single Arrhenius temperature dependence. Bulk water is found to have a greater influence on the rotational relaxation in the presence of a hydrophobic surface and the dipole angular distributions show distinct differences on the surfaces upon supercooling. Our results indicate that not only does the local extent of surface hydrophilicity play a role in determining the energy landscape explored by the water molecules upon supercooling, but the presence of bulk water also modulates the dynamic transition.


 Please wait while we load your content...
Please wait while we load your content...