Thermal stability mechanism via energy absorption by chemical bonds bending and stretching in free space and the interlayer reaction of layered molecular structure explosives†
Abstract
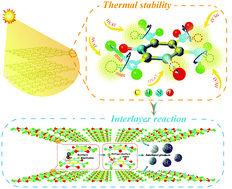
Layered molecular structure explosives have the characteristic of great thermal stability. Understanding the mechanism of thermal stability and the reactions of layered molecular structure explosives can provide new ideas for the design of thermally stable explosives. In a molecular dynamics simulation of thermal decomposition of the layered molecular structure explosive 2,4,6-triamino-5-nitropyrimidine-1,3-dioxide, we find that the layered molecular structure provides free space for chemical bond deflection and expansion so that the external energy absorbed by chemical bonds on nonbenzene rings can be converted into angle bending energy and bond-stretching energy, which makes chemical bonds difficult to break and increases the thermal stability of the explosives. In the layered molecular structure explosive reactions, hydrogen–oxygen-bonded interlayer dimerizations and hydrogen interlayer transfer reactions are dominant.


 Please wait while we load your content...
Please wait while we load your content...