Two-dimensional type-II g-C3N4/SiP–GaS heterojunctions as water splitting photocatalysts: first-principles predictions†
Abstract
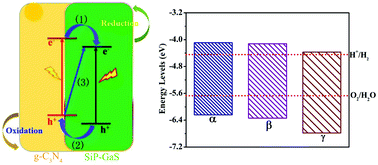
Hydrogen production from water splitting by sunlight is a promising approach to solve the increasing energy and environmental crises, and the two-dimensional (2D) g-C3N4 monolayer is a red star in this realm. However, it suffers from low quantum efficiency caused by the fast combination of photogenerated electrons and holes. In this work, we investigate the electronic and photocatalytic properties of three newly proposed g-C3N4/SiP–GaS-α, -β and -γ heterojunctions via first principles predictions. Theoretical results demonstrate that the three g-C3N4/SiP–GaS heterojunctions exhibit direct bandgaps of ∼2.2 eV, and have a type-II band alignment with the valence band maximum (VBM) located at the g-C3N4 layer and the conduction band minimum (CBM) at the SiP–GaS layer. Furthermore, their band edges straddle the redox potential of water in a wide range of biaxial strain. Their absorption coefficients are several times larger than that of most previously discovered 2D heterojunctions. Moreover, the in-built electric field adds a driving force to separate photogenerated electrons and holes. The oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) successfully take place on the g-C3N4 and SiP–GaS layers, respectively. Briefly, separated charge carriers, suitable band edges and strong visible-light absorbance, successful OER and HER enable the three g-C3N4/SiP–GaS heterojunctions to be promising water-splitting photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...