Influence of stepwise oxidation on the structure, stability, and properties of planar pentacoordinate carbon species CAl5+†
Abstract
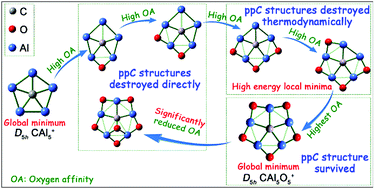
Computational design has played an important role in planar hyper-coordinate carbon (phC) chemistry. However, none of numerous computationally predicted phC species were subsequently successfully synthesized in the condensed phase, perhaps due to the frustrating issue of oxidation. In the present work, we studied the influence of stepwise oxidation on the structure, stability, and properties of phC species using the milestone planar pentacoordinate carbon (ppC) species CAl5+ as an example. Our results indicated that the ppC structure of CAl5+ would be directly destroyed with one, two, or six O atom(s) per molecule present and indirectly with three or four O atoms, but maintained with five O atoms due to the ppC isomer of CAl5O5+ being a kinetically stable global energy minimum displaying σ and π double aromaticity. Moreover, the magnitudes of the first to fifth vertical oxygen affinities (VOAs) for CAl5+ were determined to be very high (−85.5 to −116.3 kcal mol−1), probably due to the existence of peripheral diffuse Al–Al bond(s). However, the sixth VOA was reduced significantly to −50.2 kcal mol−1, consistent with the absence of any diffuse Al–Al bond in the corresponding CAl5O5+ species. So CAl5O5+ may be insensitive to oxidation. Therefore, the ppC species D5h CAl5O5+ might be resistant to being degraded under a delicate control of oxidation level (producing five O atoms per CAl5+ molecule).


 Please wait while we load your content...
Please wait while we load your content...