Stabilizing the crystal structures of NaFePO4 with Li substitutions†
Abstract
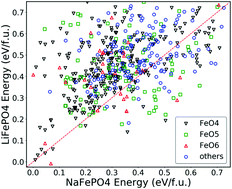
Due to the high cost and insufficient resources of lithium, alternative sodium-ion batteries have been widely investigated for large-scale applications. NaFePO4 has the highest theoretical capacity of 154 mA h g−1 among the iron-based phosphates, which makes it an attractive cathode material for Na-ion batteries. Experimentally, LiFePO4 has been highly successful as a cathode material in Li-ion batteries because its olivine crystal structure provides a stable framework during battery cycling. In NaFePO4, maricite replaces olivine as the most stable phase. However, the maricite phase is experimentally found to be electrochemically inactive under normal battery operating voltages (0–4.5 V). We found that partial substitutions of Na with Li stabilize the olivine structure and may be a way to improve the performance of NaFePO4 cathodes. Using the previously developed structural LiFePO4 database, we examined the low-energy crystal structures in the system when we replace Li with Na. The known maricite and olivine NaFePO4 phases are reconfirmed and an unreported phase with energy between them is identified by our calculations. Besides, the Li-doped olivine type compound LixNa1−xFePO4 with mixed alkali ions retains better energetic stability compared with the other two types of structures of the same composition, as long as the proportion of Li exceeds 0.25. The thermodynamic stability of o-type LixNa1−xFePO4 can be further improved at finite temperatures. The primary limitation of the calculations is that we mainly focus on the zero-temperature condition; however, the relative stability of the structures may vary depending on the ambient temperature.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...