Enhanced extraction of phenol from model oils using ionic liquids elucidated with neutron diffraction†
Abstract
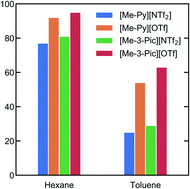
Aromatic cation ionic liquids (ILs) based on alkylpyridiniums are shown to be good phenol extractants from model oils (hexane/toluene). ILs with hard basic anions are found to have best extraction efficiency consistent with tetraalkylammonium salts ([NR4]X). Key extraction interactions were analysed using small angle neutron diffraction. Trifluoromethanesulphonate ([OTf]− or triflate) anions provide the synergistic effects of reduced cation–phenol centre of mass (COM) distances and increased hydrogen bonding that are linked to the improved extraction efficiency. Increases in cation electron density (methylpyridinium ([Me-Py]+) vs. methylpicolinium ([Me-3-Pic]+)) also reduce cation–phenol COM interaction lengths consistent with small increases in extraction efficiency for the same ionic liquids.


 Please wait while we load your content...
Please wait while we load your content...